

Yes, we know that we said helium was in group VIII, but instead of being in the p-block like all of the other elements in group VIII, it is found in the s-block. Elements are also shown with their atomic number and relative atomic mass. For example, copper is known as Cu, calcium as Ca, and carbon as C. The first letter is always capitalised and the second letter is in lowercase. This is a one or two-letter abbreviation, unique to each element. Its exact number of protons determines its number of electrons this is what makes an element, well, an element !Įlements in the periodic table are shown using their chemical symbol. Remember that an element is a pure chemical substance consisting of atoms that all have the same number of protons in their nuclei.
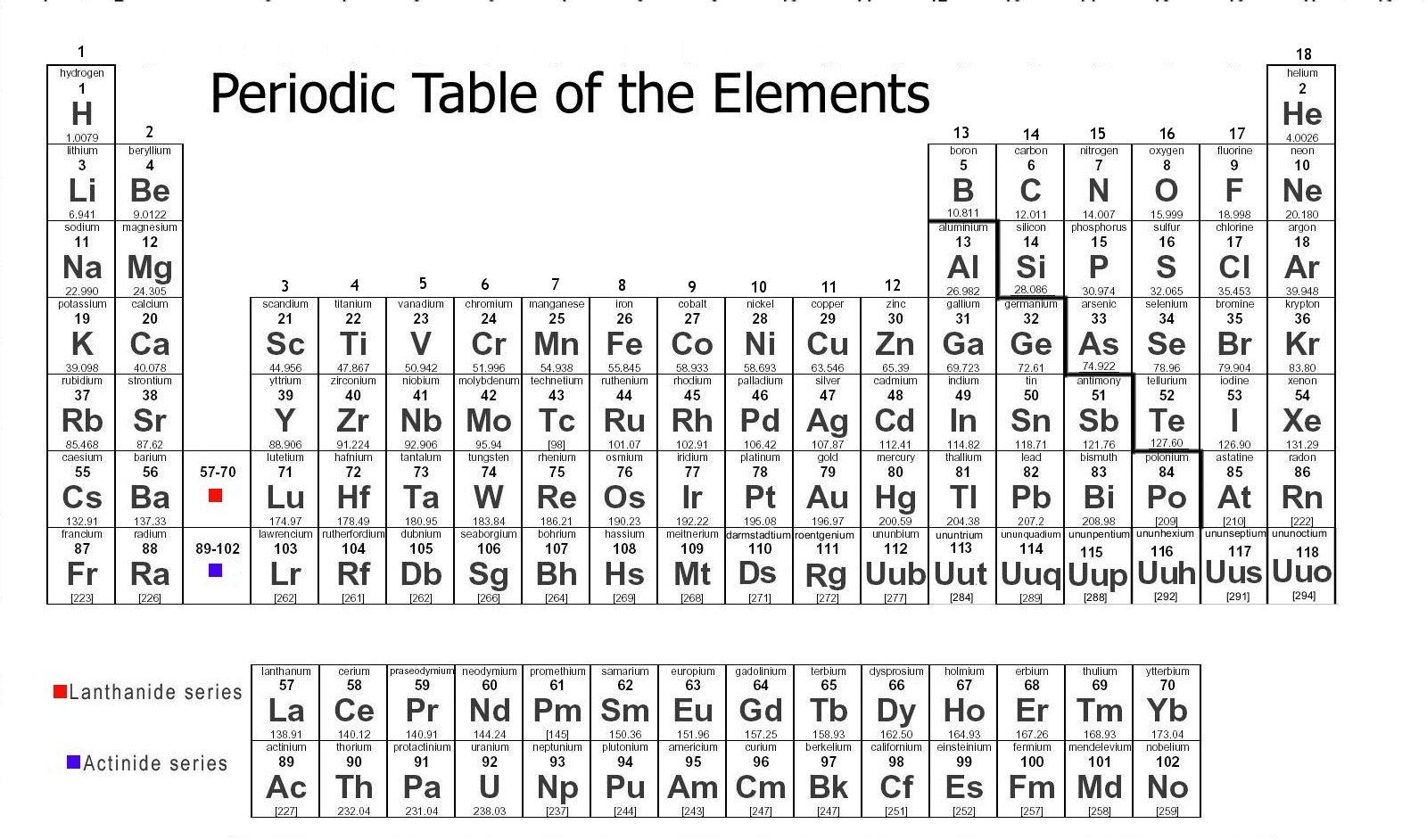
ElementsĪs we defined above, the periodic table is an arrangement of elements. As of now, we'll delve into the ways the periodic table is structured. It hasn't always been like this, but we'll get into how it has changed later, when we discuss the history of the periodic table. These elements are assembled in rows, columns, and blocks. How is the periodic table structured? First and foremost, the periodic table consists of elements. This means that once you know an element's position in the periodic table, you can predict how it behaves and reacts. Olive Odagbu, StudySmarter Originals The periodic table is handy because it orders elements into rows and columns based on their properties.
Intramolecular Force and Potential Energy.Variable Oxidation State of Transition Elements.Transition Metal Ions in Aqueous Solution.The periodic table as a list of elements arranged so as to demonstrate trends in their physical and chemical properties.Describe and model the structure of the atom in terms of the nucleus, protons, neutrons and electrons comparing mass and charge of protons neutrond and electrons. Use the Periodic Table to predict the ratio of atoms in compounds of two elements. Unit 1: Structures, Trends, Chemical Reactions, Quantitative Chemistry and Analysis.Unit C1: Structures, Trends, Chemical Reactions, Quantitative Chemistry and Analysis.(g) elements being arranged in order of increasing atomic number and in groups and periods in the modern Periodic Table, with elements having similar properties appearing in the same groups.2.2 ATOMIC STRUCTURE AND THE PERIODIC TABLE.(h) elements being arranged in order of increasing atomic number and in groups and periods in the modern Periodic Table, with elements having similar properties appearing in the same groups.1.2 ATOMIC STRUCTURE AND THE PERIODIC TABLE.Unit 1: CHEMICAL SUBSTANCES, REACTIONS and ESSENTIAL RESOURCES.(a) elements being arranged according to atomic number in the Periodic Table.Unit 1: THE LANGUAGE OF CHEMISTRY, STRUCTURE OF MATTER AND SIMPLE REACTIONS.The Periodic Table can be used to determine whether an element is a metal or non-metal.Atomic structure and bonding related to properties of materials.Elements are arranged in the periodic table in order of increasing atomic number.RSC Yusuf Hamied Inspirational Science Programme.Introductory maths for higher education.The physics of restoration and conservation.

